COMPANY NEWS
Essex and Henlius Entered into a Global Co-Development and Exclusive License Agreement to Jointly Develop Bevacizumab for Treatment of Ophthalmic Diseases
2020.10.15
Download
Hong Kong, 15 October 2020
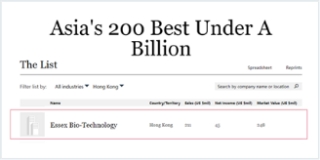
Essex Bio-Technology Ltd (“Essex” or the “Group”, Stock Code: 1061.HK) today announced that two of its wholly-owned subsidiaries, Essex Bio-Investment Limited and Zhuhai Essex Bio-Pharmaceutical Co. Ltd, entered into a global Co-Development and Exclusive license Agreement with Shanghai Henlius Biotech, Inc. (“Henlius”, Stock Code: 2696.HK) (the “Agreement”). Pursuant to the Agreement, Essex and Henlius will co-develop the bevacizumab HLX04 (the “Product”) for treatment of ophthalmic diseases such as exudative (wet) Age-related Macular Degeneration (wAMD). Essex has an exclusive global license to develop, manufacture and commercialize the Product in the field of human ophthalmic therapeutic use and/or therapies.
Pursuant to the Agreement, Henlius is mainly responsible for the pre-clinical and clinical development of the Product, whereas, Essex is responsible for regulatory filings and commercialisation of the Product globally. Essex will share the development costs in the aggregate amount of up to US$30 million with Henlius as to 80% by Essex and 20% by Henlius. If the development costs exceed US$30 million, Essex and Henlius will further negotiate on the cost sharing arrangement. In the event that Essex grants a sub-license to a third party for the commercialization of the Product, Essex and Henlius are entitled to share 80% and 20% of the sub-licensing revenues, respectively. Such sharing ratio can be adjusted through bilateral negotiation. Under the terms of the Agreement, Henlius is entitled to receive a signing payment of US$10 million, and further subsequent regulatory milestone payment including US$5 million upon initiation of the global clinical trial programme and further US$10 million upon completion of the global clinical trial programme. In addition, to reward Henlius, Essex agrees to make commercial milestone payments of up to US$18 million when the cumulative net sales generated by Essex from the Product achieve specified milestones, and US$30 million for every net sales of US$1 billion above specified milestone. Henlius is also eligible to receive royalties at a progressive rate from 6% to 10% of the annual net sales of the Product. Through this collaboration, Essex and Henlius will synergize the benefits and bear risks and costs in the development of the Product for the treatment of ophthalmic diseases. This collaboration will fully utilize the resources and expertise of Henlius in research and clinical development and Essex in global regulatory filings and commercialisation of ophthalmic drugs, to best strategize for the global development and commercialization of the Product.
The incidence of ophthalmic diseases is increasing year by year in China, especially in the middle-aged and elderly patients. As of now, the number of patients with wAMD has exceeded 5 million in China and up to 30 million worldwide [1-2]. China is currently experiencing the effects of an aging population with approximately 250 million people aged 60 and above. With an estimated 400 million people aged 60 and above by 2035 [3-4], we would expect a continued and expanded need for treatment addressing chronic and age-related ophthalmic diseases. One such potential treatment option is HLX04 (recombinant anti-VEGF humanized monoclonal antibody injection), a bevacizumab biosimilar independently developed and manufactured by Henlius. Via specific binding with vascular endothelial growth factor (VEGF), HLX04 can block the interaction between VEGF and its receptors, thus inhibiting angiogenesis. Results from pre-clinical and clinical studies have demonstrated significant efficacy of VEGF inhibitors on ophthalmic diseases caused by fundus angiogenesis [5-6]. It may be noteworthy that bevacizumab has not been approved globally for the treatment of these ophthalmic disease despite its broad use. The Product aims to be one of the first approved for use bevacizumab, addressing clinical needs and current regulatory challenges.
With the aim to benefit a broader patient population, the Co-Development Agreement is to develop the Product for ophthalmic diseases, including exudative (wet) Age-related Macular Degeneration (wAMD) and Diabetic Retinopathy (DR). In January 2019, the investigational new drug application (IND) of the Product for indications of wAMD and DR has been approved by the NMPA. With this collaboration, Essex and Henlius plan to conduct global multi-centre clinical trials of the Product in China, Australia, the European Union and the United States, and apply for marketing authorization in different countries and regions around the globe based on the research results. Meanwhile, Henlius has submitted a patent for a new formulation of the Product with potential better safety and stability, designed for ophthalmic use.
For Essex, the global partnership with Henlius on bevacizuimab for treatment of ophthalmic diseases will enrich the Group’s pipeline in ophthalmology, and strengthen the Group’s market position in ophthalmic prescribed drugs, especially in the segment of retinal disease, moving the Group one step closer to a leading ophthalmology company.
About Essex
Essex Bio-Technology Limited is a bio-pharmaceutical company that develops, manufactures and commercialises genetically engineered therapeutic rb-bFGF (FGF-2), having six commercialised biologics marketed in China since 1998. The products of the Company are principally prescribed for the treatment of wounds healing and diseases in Ophthalmology and Dermatology, which are marketed and sold through approximately 8,880 hospitals and managed directly by its 43 regional sales offices in China. Leveraging on its in-house R&D platform in growth factor and antibody, the Company maintains a pipeline of projects in various clinical stages, covering a wide range of fields and indications.
About Henlius
Henlius (2696.HK) is a global biopharmaceutical company with the vision to offer high-quality, affordable and innovative biologic medicines for patients worldwide with a focus on oncology and autoimmune diseases. Since its inception in 2010, Henlius has built an integrated biopharmaceutical platform with core capabilities of high-efficiency and innovation embedded throughout the whole product life cycle including R&D, manufacturing and commercialisation. It has three R&D facilities in Shanghai, Taipei and California and a Shanghai-based manufacturing facility certificated by China and the European Union (EU) Good Manufacturing Practice (GMP).
Henlius has pro-actively built a diversified and high-quality product pipeline covering over 20 innovative monoclonal antibodies (mAb) and has continued to explore immuno-oncology combination therapies with proprietary HLX10 (anti-PD-1 mAb) as backbone. Up to date, Henlius has launched two products, 汉利康® (HLX01, rituximab injection) , the first biosimilar approved in China and 汉曲优® (HLX02, trastuzumab injection, Zercepac® in the EU), the first Chinese mAb biosimilar launched in both China and Europe. 汉曲优® will bring more treatment options to patients with HER2 positive breast and gastric cancer worldwide. Two products (HLX03, adalimumab injection and HLX04, bevacizumab injection) were accepted for New Drug Application (NDA) review by the NMPA with HLX03 to be potentially launched within 2020. What’s more, Henlius has conducted over 20 clinical studies for 10 products and 8 combination therapies worldwide.
Reference
[1] 中华医学会眼科学分会眼底病学组中国老年性黄斑变性临床诊断治疗路径[J].中华眼底病杂志,2013,29(4):343-355.
[2] 邹海东,张皙,许迅等,上海市静安区曹家渡街道年龄相关性黄斑变性的患病率调查。中华眼科杂志,2005,41(1)15-19.
[3] 2019 Annual Data from National Bureau of Statistics (PRC)
[4] https://k.sina.com.cn/article_6192937794_17120bb420200134k2.html?from=news&subch=onews
[5] Chen C Y, Wong T Y, Heriot W J. Intravitreal Bevacizumab (Avastin) for Neovascular Age-related Macular Degeneration: A Short-term Study [J]. American Journal of Ophthalmology, 2007, 143(3):510-512.
[6] Rich R M , Rosenfeld P J , Puliafito C A , et al. Short-term safety and efficacy of intravitreal bevacizumab (Avastin) for neovascular age-related macular degeneration.[J]. Retina, 2006, 26(5):495.
Essex Bio-Technology Ltd (“Essex” or the “Group”, Stock Code: 1061.HK) today announced that two of its wholly-owned subsidiaries, Essex Bio-Investment Limited and Zhuhai Essex Bio-Pharmaceutical Co. Ltd, entered into a global Co-Development and Exclusive license Agreement with Shanghai Henlius Biotech, Inc. (“Henlius”, Stock Code: 2696.HK) (the “Agreement”). Pursuant to the Agreement, Essex and Henlius will co-develop the bevacizumab HLX04 (the “Product”) for treatment of ophthalmic diseases such as exudative (wet) Age-related Macular Degeneration (wAMD). Essex has an exclusive global license to develop, manufacture and commercialize the Product in the field of human ophthalmic therapeutic use and/or therapies.
Pursuant to the Agreement, Henlius is mainly responsible for the pre-clinical and clinical development of the Product, whereas, Essex is responsible for regulatory filings and commercialisation of the Product globally. Essex will share the development costs in the aggregate amount of up to US$30 million with Henlius as to 80% by Essex and 20% by Henlius. If the development costs exceed US$30 million, Essex and Henlius will further negotiate on the cost sharing arrangement. In the event that Essex grants a sub-license to a third party for the commercialization of the Product, Essex and Henlius are entitled to share 80% and 20% of the sub-licensing revenues, respectively. Such sharing ratio can be adjusted through bilateral negotiation. Under the terms of the Agreement, Henlius is entitled to receive a signing payment of US$10 million, and further subsequent regulatory milestone payment including US$5 million upon initiation of the global clinical trial programme and further US$10 million upon completion of the global clinical trial programme. In addition, to reward Henlius, Essex agrees to make commercial milestone payments of up to US$18 million when the cumulative net sales generated by Essex from the Product achieve specified milestones, and US$30 million for every net sales of US$1 billion above specified milestone. Henlius is also eligible to receive royalties at a progressive rate from 6% to 10% of the annual net sales of the Product. Through this collaboration, Essex and Henlius will synergize the benefits and bear risks and costs in the development of the Product for the treatment of ophthalmic diseases. This collaboration will fully utilize the resources and expertise of Henlius in research and clinical development and Essex in global regulatory filings and commercialisation of ophthalmic drugs, to best strategize for the global development and commercialization of the Product.
The incidence of ophthalmic diseases is increasing year by year in China, especially in the middle-aged and elderly patients. As of now, the number of patients with wAMD has exceeded 5 million in China and up to 30 million worldwide [1-2]. China is currently experiencing the effects of an aging population with approximately 250 million people aged 60 and above. With an estimated 400 million people aged 60 and above by 2035 [3-4], we would expect a continued and expanded need for treatment addressing chronic and age-related ophthalmic diseases. One such potential treatment option is HLX04 (recombinant anti-VEGF humanized monoclonal antibody injection), a bevacizumab biosimilar independently developed and manufactured by Henlius. Via specific binding with vascular endothelial growth factor (VEGF), HLX04 can block the interaction between VEGF and its receptors, thus inhibiting angiogenesis. Results from pre-clinical and clinical studies have demonstrated significant efficacy of VEGF inhibitors on ophthalmic diseases caused by fundus angiogenesis [5-6]. It may be noteworthy that bevacizumab has not been approved globally for the treatment of these ophthalmic disease despite its broad use. The Product aims to be one of the first approved for use bevacizumab, addressing clinical needs and current regulatory challenges.
With the aim to benefit a broader patient population, the Co-Development Agreement is to develop the Product for ophthalmic diseases, including exudative (wet) Age-related Macular Degeneration (wAMD) and Diabetic Retinopathy (DR). In January 2019, the investigational new drug application (IND) of the Product for indications of wAMD and DR has been approved by the NMPA. With this collaboration, Essex and Henlius plan to conduct global multi-centre clinical trials of the Product in China, Australia, the European Union and the United States, and apply for marketing authorization in different countries and regions around the globe based on the research results. Meanwhile, Henlius has submitted a patent for a new formulation of the Product with potential better safety and stability, designed for ophthalmic use.
For Essex, the global partnership with Henlius on bevacizuimab for treatment of ophthalmic diseases will enrich the Group’s pipeline in ophthalmology, and strengthen the Group’s market position in ophthalmic prescribed drugs, especially in the segment of retinal disease, moving the Group one step closer to a leading ophthalmology company.
About Essex
Essex Bio-Technology Limited is a bio-pharmaceutical company that develops, manufactures and commercialises genetically engineered therapeutic rb-bFGF (FGF-2), having six commercialised biologics marketed in China since 1998. The products of the Company are principally prescribed for the treatment of wounds healing and diseases in Ophthalmology and Dermatology, which are marketed and sold through approximately 8,880 hospitals and managed directly by its 43 regional sales offices in China. Leveraging on its in-house R&D platform in growth factor and antibody, the Company maintains a pipeline of projects in various clinical stages, covering a wide range of fields and indications.
About Henlius
Henlius (2696.HK) is a global biopharmaceutical company with the vision to offer high-quality, affordable and innovative biologic medicines for patients worldwide with a focus on oncology and autoimmune diseases. Since its inception in 2010, Henlius has built an integrated biopharmaceutical platform with core capabilities of high-efficiency and innovation embedded throughout the whole product life cycle including R&D, manufacturing and commercialisation. It has three R&D facilities in Shanghai, Taipei and California and a Shanghai-based manufacturing facility certificated by China and the European Union (EU) Good Manufacturing Practice (GMP).
Henlius has pro-actively built a diversified and high-quality product pipeline covering over 20 innovative monoclonal antibodies (mAb) and has continued to explore immuno-oncology combination therapies with proprietary HLX10 (anti-PD-1 mAb) as backbone. Up to date, Henlius has launched two products, 汉利康® (HLX01, rituximab injection) , the first biosimilar approved in China and 汉曲优® (HLX02, trastuzumab injection, Zercepac® in the EU), the first Chinese mAb biosimilar launched in both China and Europe. 汉曲优® will bring more treatment options to patients with HER2 positive breast and gastric cancer worldwide. Two products (HLX03, adalimumab injection and HLX04, bevacizumab injection) were accepted for New Drug Application (NDA) review by the NMPA with HLX03 to be potentially launched within 2020. What’s more, Henlius has conducted over 20 clinical studies for 10 products and 8 combination therapies worldwide.
Reference
[1] 中华医学会眼科学分会眼底病学组中国老年性黄斑变性临床诊断治疗路径[J].中华眼底病杂志,2013,29(4):343-355.
[2] 邹海东,张皙,许迅等,上海市静安区曹家渡街道年龄相关性黄斑变性的患病率调查。中华眼科杂志,2005,41(1)15-19.
[3] 2019 Annual Data from National Bureau of Statistics (PRC)
[4] https://k.sina.com.cn/article_6192937794_17120bb420200134k2.html?from=news&subch=onews
[5] Chen C Y, Wong T Y, Heriot W J. Intravitreal Bevacizumab (Avastin) for Neovascular Age-related Macular Degeneration: A Short-term Study [J]. American Journal of Ophthalmology, 2007, 143(3):510-512.
[6] Rich R M , Rosenfeld P J , Puliafito C A , et al. Short-term safety and efficacy of intravitreal bevacizumab (Avastin) for neovascular age-related macular degeneration.[J]. Retina, 2006, 26(5):495.
































































 粤公网安备 44049102496184号
粤公网安备 44049102496184号